Clinically proven. Kid-approved.
FDA authorization supported by 5 clinical studies with over 600 children with ADHD.³
Key findings
In our initial clinical trial, patients aged 8-12 played for ~25 minutes per day, 5 days per week for 4 weeks — and a more recent trial demonstrated that a second month of treatment can lead to increased benefits.

68% of parents reported improvement in ADHD-related impairments after two months of treatment.²

1 in 3 children no longer had an attention deficit on at least one measure of attention.¹

73% of children said they could more easily pay attention after one month of treatment.³
¹ Kollins et al, Lancet Digital Health 2020. *Data from a “Yes-No” Exit Survey.
² STARS-ADHD-Adjunctive: Kollins et al, npj digital medicine, 2021.
³ EndeavorRx Instructions for Use

A closer look at the data
In the STARS-ADHD* pivotal trial, EndeavorRx achieved the primary endpoint, with a mean change in the TOVA® API** (Attention Performance Index) of 0.93, significant compared to a digital control (p=0.006)*
In the STARS-Adjunct open-label study, 206 children aged 8-14 years with ADHD were studied in 2 cohorts: 1) children treated with ADHD stimulant medication (on stimulants, n=130) & 2) children not treated with any ADHD medication (no stimulants, n=76)
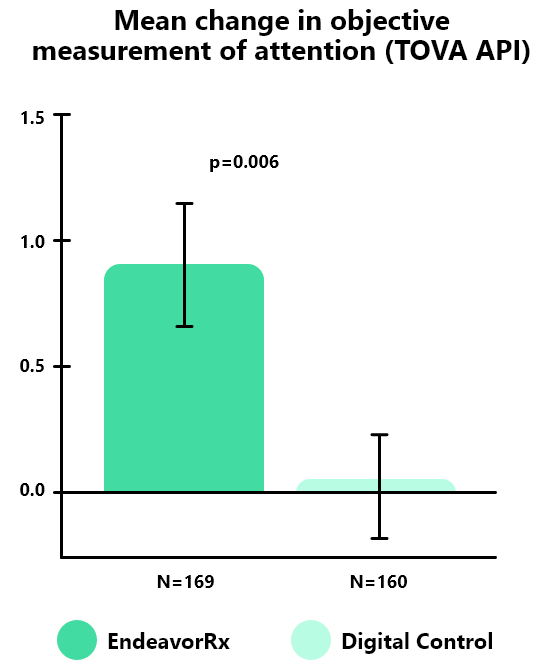
*STARS-ADHD Study Design: a randomized, double-blind, parallel-group, 4-week, controlled trial of AKL-T01 in children aged 8-12 years old with ADHD (not taking ADHD medications) and TOVA® API baseline scores of ≤ −1.8 (indicating impaired attention function) conducted at 20 sites in the USA. 348 subjects were randomly assigned to receive EndeavorRx (AKL-T01) (n=180) or digital control (n=168) for approximately 25 minutes per day, 5 days per week, for 4 weeks.
**The TOVA® – Test of Variables of Attention – is an FDA-authorized objective, computerized continuous performance test measuring attention and inhibitory control, and can aid in the diagnosis and treatment evaluation of ADHD. The TOVA® API (Attention Performance Index) (also known as TOVA® ACS: Attention Composite Score) is a composite measure that captures attentional functioning at a broad scale, including aspects of sustained and selective attention.
A meaningful impact
In the STARS-Adjunct open-label study, 206 children aged 8-14 years with ADHD were studied in 2 cohorts: 1) children treated with ADHD stimulant medication (on stimulants, n=130) & 2) children not treated with any ADHD medication (no stimulants, n=76)
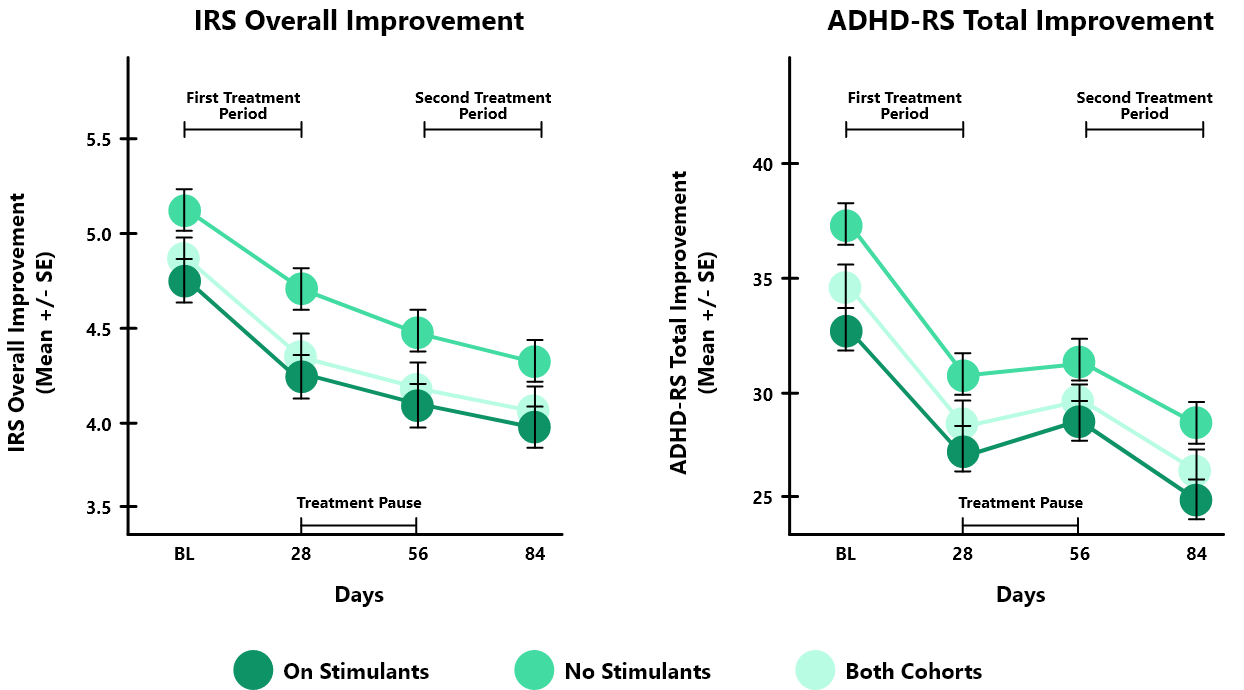
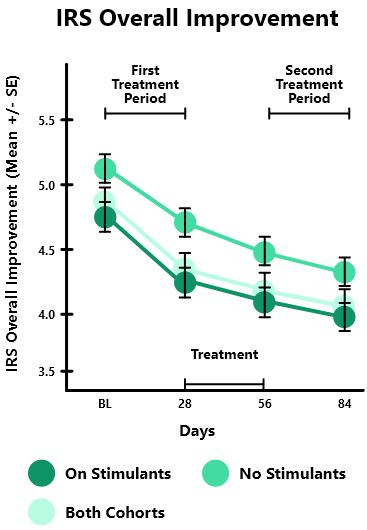
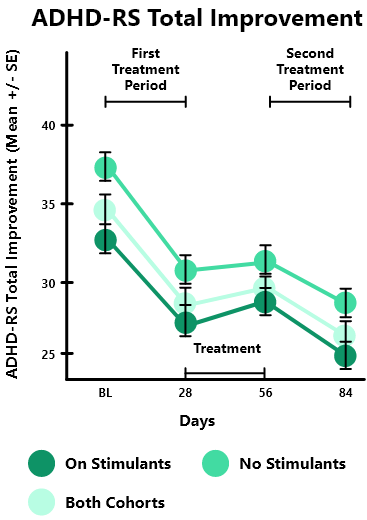
Both groups were instructed to play EndeavorRx for approximately 25 minutes per day, 5 days per week, over two 4-week treatment periods, separated by a 4-week treatment pause.
After the first 4-week treatment, IRS* overall severity score was significantly improved for both the On Stimulants (-0.7, p<0.001) and No Stimulants (-0.5, p<0.001) cohorts compared to baseline. ADHD-RS** (Total, Inattentive and Hyperactive subscales) and CGI-I were also significantly improved for both cohorts compared to baseline at day 28 and further improved with an additional treatment month (baseline to after the second 4-week treatment).
Treatment demonstrated consistent improvements in ADHD symptoms (as measured by ADHD-RS) across our 5 clinical studies, with responder rates (based on a threshold of clinically meaningful change) ranging from 23-36% for one month of treatment to 45% after two months of treatment.
Of 538 participants in trials supporting EndeavorRx authorization, 50 participants (9.3%) experienced treatment-related adverse events, most commonly frustration (6.1%), headache (1.3%), dizziness (0.6%), emotional reaction (0.4%), nausea (0.4%), and aggression (0.2%). All adverse events were generally transient and there were no serious adverse events reported.
* The IRS: Impairment Rating Scale measures parent-reported ADHD-related impairment across domains such as social functioning, academic progress, and self-esteem, including an overall impairment.
** ADHD-Rating Scale (ADHD-RS), is a ADHD symptoms rating scale. It consists of 18 questions corresponding directly to the DSM symptom criteria for ADHD and offers subscales for the assessment of inattention/hyperactivity-impulsivity.
Safety Indications
View and download our safety overviews to determine if EndeavorRx is right for your patients.


“It was really nice to see the stable benefits at her four week follow up appointment. Her academic performance, deficits in focus, and ability to sustain attention for prolonged periods – especially after school, during homework time, when stimulants wore off – had all gotten better.”
– Katherine Quinn, APRN

“When you look at attention compared to control, there’s a significant amount of symptom improvement with patients in the 8-12 group that are utilizing it.”
– Dr. Chris Dietrich, PA-C, PSY-CAQ, DSc
EndeavorRx:
Indications, Safety and Cautions
Indications:
EndeavorRx is a digital therapeutic indicated to improve attention function as measured by computer-based testing in children ages 8-17 years old with primarily inattentive or combined-type ADHD, who have a demonstrated attention issue. Patients who engage with EndeavorRx demonstrate improvements in a digitally assessed measure, Test of Variables of Attention (TOVA®), of sustained and selective attention and may not display benefits in typical behavioral symptoms, such as hyperactivity. EndeavorRx should be considered for use as part of a therapeutic program that may include clinician-directed therapy, medication, and/or educational programs, which further address symptoms of the disorder.
Safety:
No serious adverse events were reported. Of 342 participants who received AKL-T01 in the two clinical trials supporting EndeavorRx authorization for age ranges 8-17, 17 participants (4.97%) experienced treatment-related adverse events (TE-ADE) (possible, probable, likely). TE-ADEs reported at greater than 1% across the studies include: frustration tolerance decreased (2.34%) and headache (1.17%). Other adverse events occurred less than 1% and included dizziness, emotional disorder, nausea, and aggression. All adverse events were transient and no events led to device discontinuation. Across other studies in children and adolescents with ADHD, rates of adverse events were similarly low (<10%) and no Serious Adverse Events have been reported. All reported adverse events across all clinical trials resolved at the end of treatment. Users should consider the totality of evidence presented along with their health care provider when considering incorporating AKL-T01 into their treatment plan.
Cautions:
Rx only: Federal law restricts this device to sale by or on the order of a licensed health care provider. EndeavorRx should only be used by the patient for whom the prescription was written. For medical questions, please contact your child’s healthcare provider. If you are experiencing a medical emergency, please dial 911. EndeavorRx is not intended to be used as a stand-alone therapeutic and is not a substitution for your child’s medication.
If your child experiences frustration, emotional reaction, dizziness, nausea, headache, eye-strain, or joint pain while playing EndeavorRx pause the treatment. If the problem persists contact your child’s healthcare provider. If your child experiences a seizure stop the treatment and contact your child’s healthcare provider.
EndeavorRx may not be appropriate for patients with photo-sensitive epilepsy, color blindness, or physical limitations that restrict use of a mobile device; parents should consult with their child’s healthcare provider.
Please follow all of your mobile device manufacturer’s instructions for the safe operation of your mobile device. For example, this may include appropriate volume settings, proper battery charging, not operating the device if damaged, and proper device disposal. Contact your mobile device manufacturer for any questions or concerns that pertain to your device.